 Our calculator provides steps so that you will get more familiar with the steps. If you want to know the step-by-step process of balancing a chemical equation then you can directly click on it. Check the equation thoroughly to know that it is perfectly balanced. Our chemical equation balancer contains a "Calculate" button. But before moving further make sure you review your chemical equation. Now start solving the unbalanced equation. You will type your unbalanced equation or chemical formulas directly. Now enter your unbalanced chemical equation into the input field. It must have a space to enter your equation which you will find easily. 
Take a moment to make yourself familiar with the chemical equations and balancing tool’s interface. You can directly enter into the website by clicking on the website’s URL. 
Find our balancing chemical equations solver. 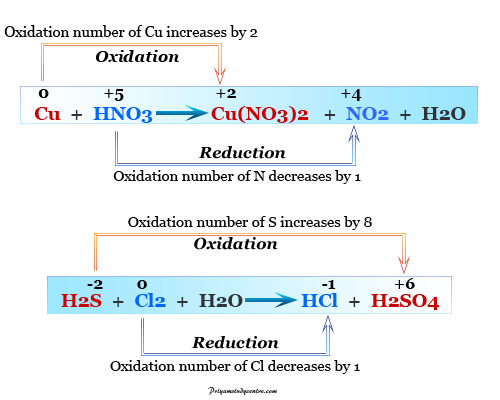
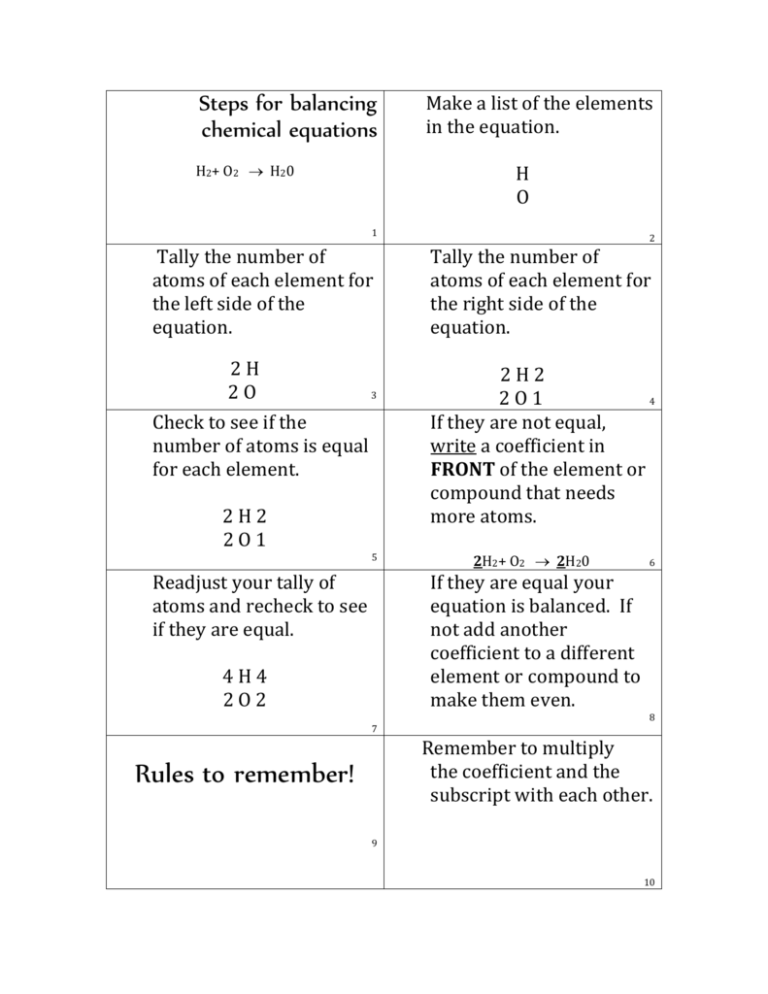
We are going to tell you easy ways to put the equation in a chemical equation solver: Find our Chemical Equation Solver:įirst of all, open your browser. Putting an equation in a chemical equation balancer is a straightforward process. $$ Zn + 2HCL → ZnCl_2 + H_2 $$ How to Put an Equation in a Balance Equation Solver? Now replace the coefficients into the chemical reaction to get a balanced chemical equation then you will get, For example, c1 = 1 and solve the equation for coefficients of both sides: Now balance the number of atoms in the reactants to the number of atoms in the products for Zinc ( Zn ), Hydrogen (H), and Chlorine (Cl). $$ CH_3 (CH_2)_3 CH_3 + 8O_2 → 5CO_2 + 6H_2O $$Įxample 2: Balance the following chemical equation:Īdding stoichiometric coefficients ( ci) on both sides, Now replace the coefficients with a chemical reaction to get a balanced chemical equation, For example, c1 = 1, and then solve the equation for the coefficients: Now equalize the number of atoms in the reactants to the number of atoms in the products for Carbon ( C ), Hydrogen (H), and Oxygen (O). $$ CH_3 (CH_2)_3 CH_3 + O_2 → CO_2 + H_2O $$Īdding stoichiometric coefficients ( ci)on both sides, To get more familiarity with balancing equations balance the following chemical equations:Įxample 1: Consider the following unbalanced chemical equation:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
