Department of Health and Human Services Office of the Inspector General (HHS-OIG). “Ensuring patient safety is our number one priority,” said Special Agent in Charge Maureen R. Jude no longer sold the older devices, but thousands of them had been implanted into patients between Nov. A Class I recall is where there is a reasonable probability that “violative” products “will cause serious adverse health consequences, including death.” After the recall, St. Jude issued a medical advisory regarding the PBD caused by lithium cluster shorts, which FDA classified as a Class I recall. 
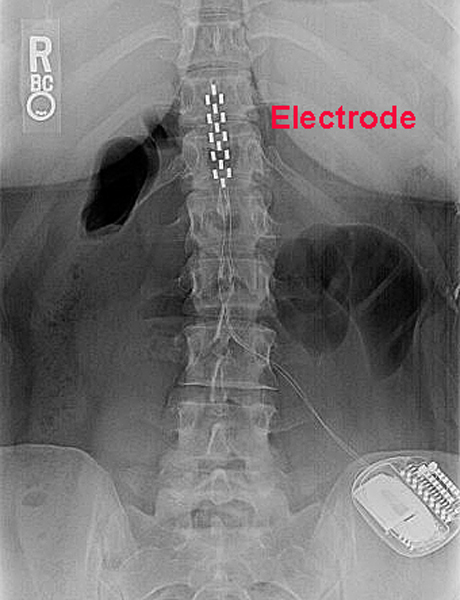
Jude contacted the FDA and informed it that the number of PBD events had increased to 729, including two deaths and 29 events associated with loss of pacing. Jude continued to distribute devices that had been manufactured without the new design. Jude was aware at that time of two reported serious injuries and one death associated with premature battery depletion (PBD) induced by lithium clusters. However, according to the government’s allegations, St. Jude submitted a request to the FDA to approve a change to prevent lithium clusters from draining the battery and told the FDA, “no serious injury, permanent harm or deaths have been reported associated with this” issue. The government alleges that, in late 2014, St. Jude knew that lithium clusters formed on the batteries of the devices, causing some of the batteries to short and, in turn, suffer a premature power drain. The government alleged that, by 2013, St. The devices are surgically implanted into patients’ chests, and when the devices detect an irregular heartbeat, they send an electrical pulse to the heart to “shock” it back to its normal rhythm. Jude failed to disclose serious adverse health events in connection with the premature depletion of the battery in certain models of its Fortify, Fortify Assura, Quadra and Unify devices, which are implantable defibrillators used in patients at risk of cardiac arrest due to an irregular heartbeat. Attorney’s Office is committed to protecting Medicare and other federal health care programs from fraud, and in doing so strengthen patient safety.” 
Jude knowingly caused the submission of false claims and failed to inform the FDA with critical information about prior injuries and a death which, had the FDA been made aware, would have led to a recall. government will not pay for devices that are unsafe and risk injury or death,” said Acting U.S. “Medical device manufacturers have an obligation to be truthful with the Food and Drug Administration (FDA), and the U.S. “We will hold accountable those companies whose conduct violates the law and puts patients’ health at risk.” Boynton of the Justice Department’s Civil Division. “To ensure the health and safety of patients, manufacturers of implantable cardiac devices must be transparent when communicating with the government about safety issues and incidents,” said Acting Assistant Attorney General Brian M. Jude was acquired by Abbott Laboratories in January 2017. Jude) has agreed to pay $27 million to settle allegations under the False Claims Act that, between November 2014 and October 2016, it knowingly sold defective heart devices to health care facilities that, in turn, implanted the devices into patients insured by federal health care programs. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
